
Routes of exposure: The substance can be absorbed into the body by inhalation. Explosion: Gas/air mixtures are explosive. Many reactions may cause fire or explosion. Hydrogen reacts with oxygen to form water and this reaction is extraordinarily slow at ambient temperature but if it’s accelerated by a catalyser, like platinum, or an electric spark, it’s made with explosive violence.Įffects of exposure to hydrogen: Fire: Extremely flammable. The heat released when the hydrogen atoms recombine to form the hydrogen molecules is used to obtain high temperatures in atomic hydrogen welding. Atomic hydrogen produces hydrogen peroxide, H 2O 2, with oxygen.Ītomic hydrogen reacts with organic compounds to form a complex mixture of products with etilene, C 2H 4, for instance, the products are ethane, C 2H 6, and butane, C 4H 10. It reacts with a number of elements, metals and non-metals, to produce hydrides, like NAH, KH, H 2S and PH 3. It reduces some salts to their metallic state, like nitrates, nitrites and sodium and potassium cyanide. It reacts with the oxides and chlorides of many metals, like silver, copper, lead, bismuth and mercury, to produce free metals. Atomic hydrogen is a powerful reductive agent, even at ambient temperature. At high temperatures it’s highly reactive.Īlthough in general it’s diatomic, molecular hydrogen dissociates into free atoms at high temperatures. Hydrogen absorption by steel can result in brittle steel, which leads to fails in the chemical process equipment.Īt normal temperature hydrogen is a not very reactive substance, unless it has been activated somehow for instance, by an appropriate catalyser. Hydrogen is slightly more soluble in organic solvents than in water. Hydrogen is the most flammable of all the known substances. Its relative density, compared with that of the air, is 0.0695. As a gas it has a density of 0.071 g/l at 0✬ and 1 atm. Properties: Common hydrogen has a molecular weight of 2,01594 g. For instance it can be converted to and from electricity from bio-fuels, from and into natural gas and diesel fuel, theoretically with no emissions of either CO2 or toxic chemicals. Hydrogen fuel cells are being looked into as a way to provide power and research is being conducted on hydrogen as a possible major future fuel. Hydrogen can be burned in internal combustion engines. Huge quantities of hydrogen are used as rocket fuels, in combination with oxygen or fluor, and as a rocket propellent propelled by nuclear energy. Hydrogenation is used in the manufacture of organic chemical products. Huge quantities of hydrogen are consumed in the catalytic hydrogenation of unsaturated vegetable oils to obtain solid fat. The use of hydrogen is extending quickly in fuel refinement, like the breaking down by hydrogen (hydrocracking), and in sulphur elimination. Uses: The most important use of hydrogen is the ammonia synthesis. There are three hydrogen isotopes: protium, mass 1, found in more than 99,985% of the natural element deuterium, mass 2, found in nature in 0.015% approximately, and tritium, mass 3, which appears in small quantities in nature, but can be artificially produced by various nuclear reactions. It’s one of the main compounds of water and of all organic matter, and it’s widely spread not only in The Earth but also in the entire Universe. 
Its atomic number is 1 and its atomic weight 1,00797 g/mol. The hydrogen atom, symbol H, is formed by a nucleus with one unit of positive charge and one electron. In normal conditions it’s a colourless, odourless and insipid gas, formed by diatomic molecules, H 2. Source: /dtrapp/Elements/properties.htmlįirst element in the periodic table. * Hydrogen was observed and collected long before it was recognized as a unique gas by Robert Boyle in 1671, who dissolved iron in diluted hydrochloric acid. Hydrogen - H Chemical properties of hydrogen - Health effects of hydrogen - Environmental effects of hydrogen Separation and Concentration Purification Request. 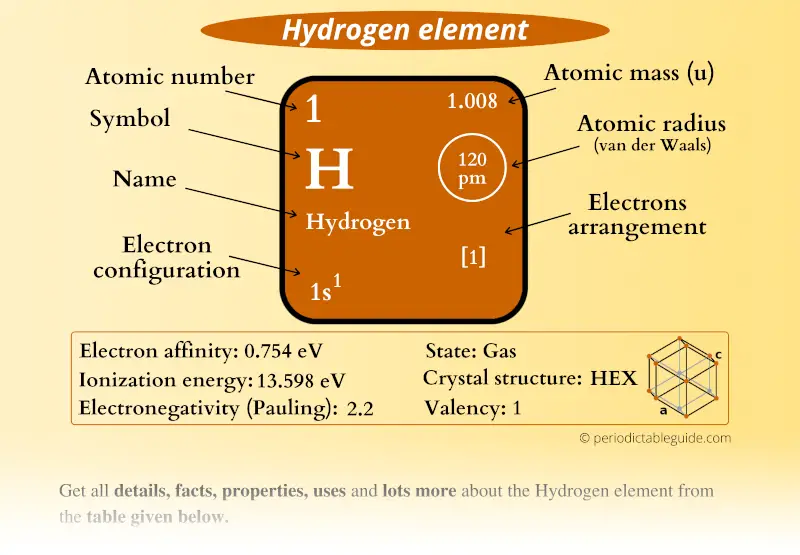
Plant Inspection & Process Optimalisation.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
